Sourcing magnets for medical devices is tough. You face strict compliance and performance demands. A wrong choice can mean project failure, budget overruns, and even safety risks for patients.
Choose medical-grade NdFeB magnets by verifying biocompatibility (ISO 10993), ensuring proper encapsulation for the application (e.g., hermetic sealing for implants), confirming sterilization compatibility, and validating MR safety according to ASTM standards. This ensures both performance and regulatory compliance for your device.

I've worked with many clients like Reza from Iran and Charalampos from Cyprus. They are brilliant engineers and sharp procurement managers, but the specific requirements for medical-grade magnets can be a maze. The stakes are incredibly high. I've seen firsthand how a small mistake in magnet specification can lead to a device failing validation or being rejected by regulators. This can set a project back by months and cost a fortune. You need to get this right from the start. Let’s break down the critical factors you need to master, step-by-step, so you can source with confidence and protect your project.
How Do You Ensure a Magnet is Safe for Patient Contact?
Your device might touch a patient, but standard NdFeB magnets contain iron and will corrode. This can release toxic materials. You need a solution that is both powerful and perfectly safe.
Ensure safety by choosing the right coating or encapsulation. For skin contact, Parylene C or gold over a nickel barrier (Ni-Cu-Ni) is common. For long-term implants, you must specify true hermetic sealing1, like a laser-welded titanium2 or ceramic case, to prevent any contact with body fluids.
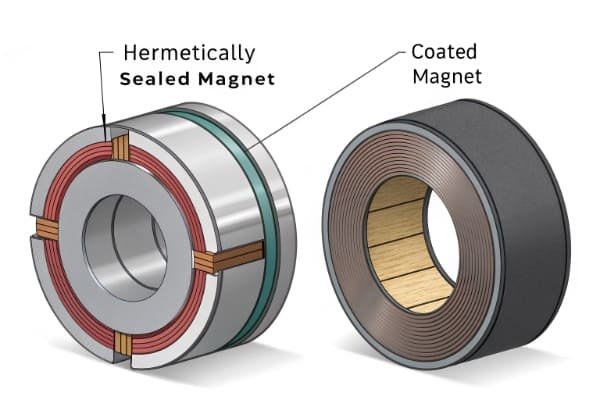
The core problem is simple: NdFeB magnets will rust if they are exposed to moisture, saline, or body fluids. This isn't just an appearance issue. The corrosion can degrade the magnet's performance and, more importantly, release materials that are not biocompatible, posing a direct risk to the patient. For any medical application, the magnet must be completely isolated. I've seen clients try to cut corners here, and it always ends badly. You have two main paths to ensure safety, and choosing the wrong one for your application is a critical error.
Coating vs. Hermetic Sealing
The choice between a simple coating and a full hermetic seal depends entirely on how your device will be used. Don't let a supplier tell you a standard coating is "good enough" for an implant; it's not.
| Solution Type | Recommended Materials | Best For | Key Requirement for Your PO |
|---|---|---|---|
| Biocompatible Coating | Parylene C3, Gold (over a Ni-Cu-Ni base) | Skin-contact, short-term invasive devices. | Specify coating material and thickness. Demand coating integrity reports and salt spray test data4. |
| Hermetic Sealing | Titanium (e.g., Ti-6Al-4V), Ceramic | Long-term implants (e.g., pacemakers, drug pumps). | Specify laser-welded assembly. Crucially, you must demand a Helium leak test5 report with a rate of ≤1×10⁻⁹ atm·cc/s. |
Relying on standard nickel plating (Ni-Cu-Ni) alone for any long-term internal device is a mistake I see too often. While it provides corrosion resistance for industrial applications, this plating is inherently porous and will eventually fail when exposed to body fluids over time. Insist on the right level of protection for your specific application.
Will Sterilization Damage Your Medical Magnet?
Your medical device must be sterilized before use. But common sterilization methods involve harsh heat, chemicals, or radiation. You're right to worry that this process will damage the magnet or its coating.
Yes, it absolutely can. Each method (EtO, Gamma, E-beam, Autoclave/Steam) has a different impact. You must specify a magnet and coating system that has been validated for your exact sterilization method. For instance, the high heat and moisture of a steam autoclave will destroy most standard coatings.
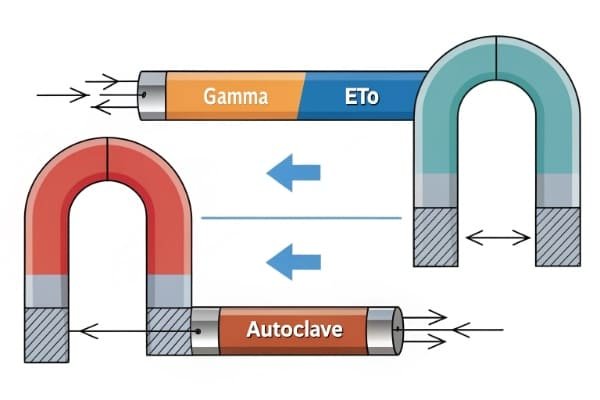
Sterilization isn't just about making things clean; it's a harsh industrial process that can wreak havoc on sensitive components like NdFeB magnets. The high temperatures can permanently reduce a magnet's strength (demagnetization), and both heat and moisture can cause coatings to crack, peel, or blister, leading to corrosion. I once helped a client whose product kept failing quality control right after it came out of sterilization. We investigated and found their supplier was providing a magnet with a standard black epoxy coating. It looked great, but it couldn't survive the steam autoclave process my client was using. The coating was failing, and the magnets were starting to corrode. The solution was simple once we identified the problem: we switched to a magnet with a more robust Parylene coating that was validated for their process. This is why you must match the magnet to the sterilization method.
Impact of Common Sterilization Methods
Don't just ask a supplier, "Is this magnet okay for medical use?". Ask them, "Have you validated this magnet with Parylene C3 coating for 5 cycles of steam autoclaving at 134°C?" Be specific.
| Method | Potential Impact on Magnet/Coating | What to Ask Your Supplier |
|---|---|---|
| EtO (Ethylene Oxide)6 | Generally safe for most magnets and coatings. | "Confirm your coating materials are compatible with EtO gas and the specified temperature/humidity cycles." |
| Gamma / E-beam7 | Radiation can degrade some polymer coatings (e.g., epoxy) and may slightly affect magnetic properties over multiple cycles. | "Can you provide data on magnetic performance and coating integrity after X kGy of gamma irradiation?" |
| Steam (Autoclave) | High Risk. The heat can demagnetize the magnet if the grade is not correct. Moisture will attack and destroy most standard coatings. | "Which high-temperature grade and coating/encapsulation do you recommend for autoclaving at X degrees?" |
Your supplier must be able to provide data to back up their claims. If they can't, find one who can.
Can Your NdFeB-based Device Be Used in an MRI?
You want your medical device to be safe for patients who may need an MRI scan. However, you know that the powerful magnetic fields of an MRI can interact dangerously with magnets, potentially pulling the device or causing it to heat up.
It depends. A device containing an NdFeB magnet is typically labeled "MR Conditional," never "MR Safe." This means it is safe only under a specific set of conditions (e.g., MRI field strength, scan duration). You must validate this safety according to a strict set of ASTM standards8.

This is one of the most misunderstood areas I discuss with clients. The terminology is critical. A paperclip is "MR Unsafe." A plastic ruler is "MR Safe." Your device, containing a powerful magnet, falls into the middle category: "MR Conditional." This means you, the device manufacturer, must define the exact conditions under which it is safe. Getting this wrong can have severe consequences. To get it right, you need to work with a supplier and a testing lab to validate performance against several key ASTM standards8. Don't just take a supplier's word for it; the final responsibility for labeling is yours.
Key Tests for MR Conditional Validation
These tests are non-negotiable for any device intended for use in an MRI environment. They directly measure the risks involved.
-
ASTM F20529: Magnetically Induced Displacement Force This test answers the question: Is the MRI's static magnetic field strong enough to pull or move the device? The force must be less than the force of gravity on the device.
-
ASTM F221310: Magnetically Induced Torque This test answers: Will the MRI's magnetic field try to twist or rotate the device out of position? This is especially critical for implants.
-
ASTM F2182: Radiofrequency (RF) Heating During a scan, the MRI emits RF energy. This test measures how much the magnet and any conductive components in your device will heat up. Excessive heating can cause tissue damage.
-
ASTM F2119: Image Artifacts The magnet in your device will create a "blind spot" or distortion in the MRI image. This test quantifies the size of that artifact, so radiologists know the limits of the scan.
As a procurement manager or engineer, you need to ensure these tests are budgeted for and performed correctly. Your magnet supplier should be able to provide magnets with consistent properties to ensure your test results are repeatable.
How Do You Verify Your Supplier's Medical Magnet Quality?
You've specified the perfect magnet on paper, but how can you trust your supplier to deliver it consistently with every shipment? You've likely been burned before by poor communication, delayed shipments, and questionable quality certificates.
Demand objective evidence for every batch. A reliable medical magnet supplier must provide a detailed Certificate of Analysis (COA)11, coating and sealing test reports (like helium leak rates), full batch traceability12, and a pre-agreed Acceptable Quality Limit (AQL)13 inspection plan. Don't accept promises.

This is where the real work begins for procurement managers like Reza and Charalampos. The pain points are always the same: inefficient communication, suppliers who don't understand the technical needs, and the constant fear of a quality issue halting production. I remember a client whose entire production line was stopped because a shipment of magnets failed their incoming inspection. The coating was inconsistent, and they couldn't risk using them. The financial loss and project delays were huge. This is why a simple "trust me" from a supplier is worthless. You need a rigorous verification process.
Your Essential Procurement Checklist
Use this checklist when you audit a new magnet supplier or create a quality agreement. This is how you move from hoping for quality to ensuring it.
- [ ] ISO 13485 Certification: ISO 9001 is for general manufacturing. ISO 13485 is the standard for medical device quality management systems. A supplier with ISO 13485 understands the documentation and traceability you require.
- [ ] Batch-Specific Certificate of Analysis (COA)11: Does the COA detail the magnetic properties (Br, Hcj) and dimensional measurements for your specific batch? A generic certificate is a red flag.
- [ ] Coating/Sealing Verification Reports: For coatings, this means reports on thickness and adhesion (e.g., cross-hatch test14). For hermetic seals, insist on the helium leak test report for that batch.
- [ ] Full Batch Traceability: Ask the supplier to demonstrate how they can trace your finished magnet all the way back to the raw material powder lot. This is non-negotiable for medical devices.
- [ ] Agreed AQL and Inspection Plan: Before you place the order, agree on the AQL level (e.g., AQL 0.65), the inspection methods, and the process for handling a rejected lot. This prevents disputes later.
By demanding this level of documentation, you force your supplier to demonstrate their quality control, and you give your own quality team the data they need to approve incoming material with confidence.
Conclusion
Sourcing medical magnets is complex, but not impossible. It requires a systematic approach focusing on biocompatibility, sterilization, MR safety, and rigorous supplier verification. Partner with an expert manufacturer to succeed.
Discover how hermetic sealing protects sensitive components in medical devices. ↩
Learn about the advantages of using laser-welded titanium for long-term implants. ↩
Find out why Parylene C is a preferred coating for medical applications. ↩
Understand the significance of salt spray tests in evaluating coating durability. ↩
Discover how Helium leak tests ensure the integrity of hermetic seals in medical devices. ↩
Learn about the effects of EtO sterilization on medical device materials. ↩
Explore the implications of radiation sterilization on medical device performance. ↩
Explore ASTM standards to ensure compliance and safety in medical device manufacturing. ↩
Learn about the ASTM F2052 test for assessing magnetic displacement in MRIs. ↩
Discover how ASTM F2213 measures the torque effects of MRI on devices. ↩
Learn about the importance of COAs in ensuring quality in medical devices. ↩
Discover the importance of batch traceability for ensuring quality and safety. ↩
Understand how AQL helps maintain quality standards in medical device production. ↩
Learn about the cross-hatch test for assessing coating adhesion in medical devices. ↩
